|
|
1.IntroductionOrganic light-emitting diodes (OLEDs) have attracted considerable attention in both academic and industrial circles since the pioneering work by Tang and Vanslyke in 1987.1 Because of advantageous properties such as flexibility, self-emission, full-color emission, low driving voltage, and fast response time, OLEDs have recently been developed and applied extensively in the mobile phone market, and many studies are underway for their applications in television and lighting.2–5 Certain properties of OLEDs make them especially attractive in the lighting market, including area emission characteristics not found for other existing light sources, environmentally friendly efficient use of energy, large area, ultra-light weight, and ultra-thin shape. It has thus become increasingly important to carry out investigations of the applications of OLEDs if they are to become the next-generation source of lighting and replace existing fluorescent and incandescent lamps.6 White OLEDs can be divided into those that produce white light by combining red light, green light, and blue light (R, G, B) emitters7–9 and those that produce white light by combining emitters of sky-blue light and orange light or of sky-blue light and red light.10–12 Investigations have been more actively pursued for the latter two-color white OLEDs whose structures are relatively simple, because low cost is regarded as important in the lighting field, unlike in the display field. In addition, since the emission spectra of organic materials generally have peaks with broader wavelengths than do the emission spectra of inorganic materials, only two types of organic emitters, in particular the emitters of sky-blue and orange light or of sky-blue and red light, need to be combined to produce white light when using OLEDs. Both fluorescent and phosphorescent materials have been investigated as emitters for use in two-color white OLEDs. The fluorescent materials that have been investigated consist of polycyclic aromatic hydrocarbons (PAHs), which are advantageous for manufacturing long-lifetime devices because of their superior stability, but have a relatively poor, at best 25%, quantum efficiency since they only use singlet excitons. In order to achieve more than a 5% external quantum efficiency (EQE), which is regarded as a limitation value, there has recently been an important research approach using a thermally activated delayed fluorescence (TADF) effect. The TADF effect is explained as an effect that allows for simultaneous use of singlet and triplet excitons in a fluorescence material by returning triplet excitons to a singlet exciton using a thermal activation process. As a representative example of a blue emitter, the Adachi group13 reported bis[4-(3,6-dimethoxycarbazole)phenyl]sulfone (DMOC-DPS), which shows the TADF effect. The photoluminescence quantum yield (PLQY) of DMOC-DPS was 0.56 in the solution state and 0.80 in the film state. In a doped device, DMOC-DPS showed a luminance efficiency (LE) of and a high EQE of 14.5%. Although the TADF series exhibits excellent characteristics, studies on TADF are still in the early stage and few papers have been presented so far. These materials were skipped in this review as it was not applied to white OLED for lighting in a full scale. Since many people are interested in these materials, it will be separately reviewed in the future. In contrast, the phosphorescent materials that have been investigated are advantageous for the manufacture of high-efficiency devices since they can harvest both singlet and triplet states and can hence theoretically achieve an internal quantum efficiency of 100%. However, these materials are relatively unstable and are thus disadvantageous with regard to device lifetime. Therefore, instead of focusing on two-color white OLEDs that consist of only fluorescent materials or only of phosphorescent materials, recent studies are more actively focusing on white OLEDs that use both fluorescent and phosphorescent emitters and hence combine the advantages of the two materials. In order to embody white OLEDs with high efficiency and long lifetime, emitters should have good thermal properties and high electroluminescence (EL) efficiency as well as a long lifetime. In order to provide a fundamental understanding of emitting materials applicable to OLED lighting, and to help direct future research in this field, this article categorizes materials that are fluorescent and those that are phosphorescent and reviews information on the characteristics of each of these materials and the devices in which they can be used. 2.Fluorescent Materials for Two-Color White OLEDsAs mentioned above, materials that emit blue light (also referred to here as blue light-emitting materials) are mainly developed from fluorescent materials based on chromophores that consist of polycyclic aromatic hydrocarbons. In order to influence the electron density of the molecules, various electron-push or -pull functional groups can be introduced into the PAH chromophore, which lengthens or shortens the wavelength of the emission. Studies on materials that emit yellow, orange, and red light are also being conducted in addition to those that emit blue light by tuning molecular characteristics or adjusting the emission wavelengths of molecules through similar methods. High-efficiency fluorescent materials used in white OLEDs generally display characteristics that, as described below, arise from three structural features: their fused and rigid aromatic rings, noncoplanar -conjugated molecular backbones, and bulky electron-donor or electron-acceptor functional groups. Fused and rigid aromatic rings are extensively employed as the emission building blocks of the chromophores, especially for materials that emit blue light, because these rings confer an inherently high PLQY and excellent thermal stability on the chromophore. They do, however, have serious drawbacks, such as tending to cause the chromophore to crystallize, as well as fluorescence quenching when used in OLED-emitting materials. To overcome these drawbacks, noncoplanar -conjugated molecular backbones are commonly utilized. These molecular backbones act as a key scaffold that effectively restricts intermolecular aggregation and thus prevents fluorescence quenching induced by aggregation and the excimer formation of the rigid and planar emitting materials in the solid film state. These are directly responsible for the ability of the chromophore to acquire high PLQYs, high EQEs, and high purity of the emission color. In addition, the characteristic noncoplanar structure also plays an important role in enhancing the stability of the amorphous glass state of the molecules, which resists crystallization when the device is being operated and affects the lifetime of the device. When designing the noncoplanar chemical structure, sterically bulky side or end-capped groups are, in many cases, intentionally incorporated into the emissive core units, which not only generate the large torsional angles but also severely interrupt intermolecular interactions between adjacent molecules. Importantly, the mutual lack of interference between the core and side groups leads to the localization of excitons solely onto the core emissive site, and not onto the whole molecular backbone, which prevents deterioration on the purity of the color of the light emitted by the core units. Finally, the bulky electron-donor or electron-acceptor functional groups are tactically introduced mostly into the core group in order to optimize the highest occupied molecular orbital (HOMO) / lowest unoccupied molecular orbital (LUMO) energy levels of the emitting materials for balancing charge transport and confinement in the emitting layer (EML) of the OLED. In this section, based on an exhaustive survey of the recently published papers, we systematically describe representative fluorescent materials that emit blue and red light for white OLEDs, with an emphasis on describing the characteristic structural properties of these materials that allow them to achieve high EQEs. The detailed EL performances for these materials are summarized in Table 1. Table 1Summary of the photoluminescence and electroluminescence performance for the emitters according to the sequence of the International Commission on Illumination (CIE) y value.
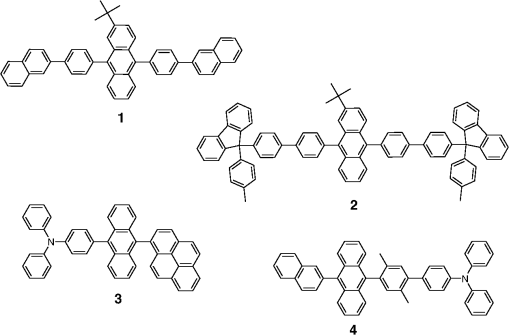
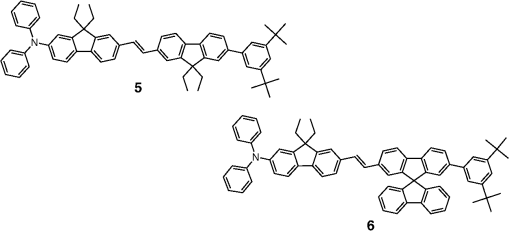
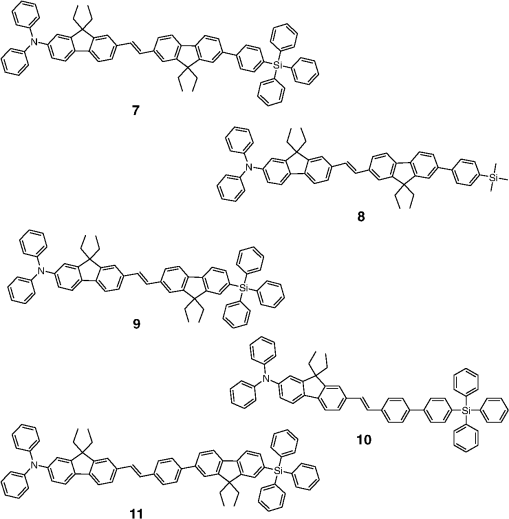
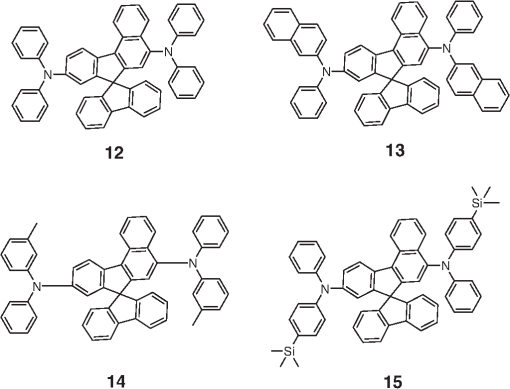
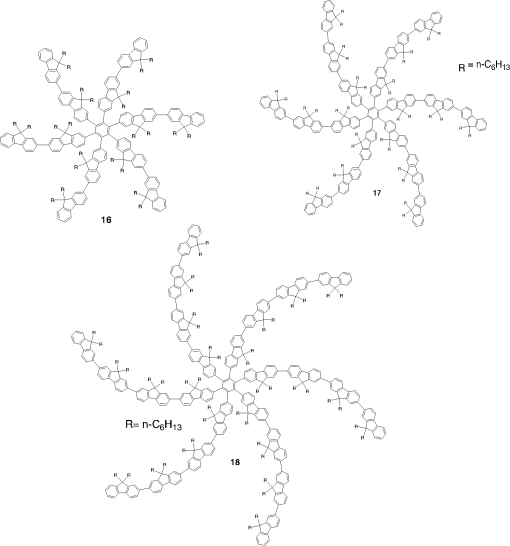
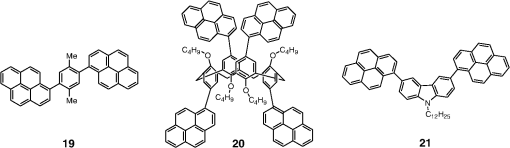
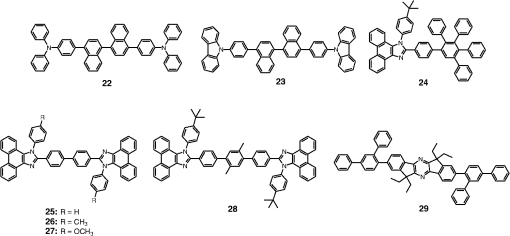
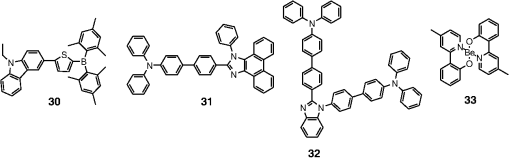
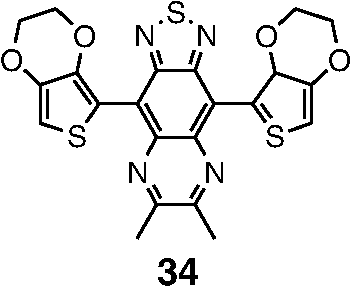
Note: LE, luminance efficiency; PE, power efficiency; EQE, external quantum efficiency. 2.1.Blue Fluorescent EmittersMaterial that emits blue light is required to investigate white OLED lighting. It has been a struggle to achieve high-performance properties for blue fluorescent materials because of the intrinsically wide differences between their HOMO and LUMO energy levels; these wide band gaps make hole injection more difficult, and electron charges migrate from the adjacent hole and electron transporting layers into the EMLs, which leads to low EQEs. Increasingly intensive and comprehensive efforts are thus being made to develop high-performance fluorescent materials that emit blue light. Among the various materials developed in this effort, blue light-emitting polycyclic, donor/acceptor and organometallic materials have prominently demonstrated highly efficient EL properties with a high quality of the blue colors of the emitted light. 2.1.1.Polycyclic-type emittersPAHs (or fused -conjugated rings), such as anthracene, fluorene, and pyrene, have been widely employed as building blocks for the design of fluorescent materials that emit blue light due to their excellent photoluminescent and thermal properties as well as wide band gaps. An increasingly larger number of sophisticated attempts have recently been undertaken to refine these polycyclic emitters for the purpose of further improving their EQEs. AnthraceneAmong these PAH molecules, anthracene derivatives have been intensively investigated for use as blue light-emitting fluorescent materials in high-performance OLEDs. In particular, 9, 10-diphenylanthracene (DPA) functionalities have been considered as the most attractive molecular skeleton to develop high-performance blue light-emitting materials because the nonplanar structure compared to anthracene itself resulting from the end-capped phenyl rings at the 9- and 10- positions of the central anthracene unit efficiently suppresses the intermolecular interactions leading to photoluminescence (PL) quenching by concentrating the emitting materials in the film state. Unfortunately, however, DPA itself tends to be crystallized during device operation, which could produce rough surfaces and grain boundaries, leading in turn to problems such as pin holes and current leakages, and ultimately catastrophic device failure. In order to improve the amorphous behaviors of DPA molecules, Zheng et al.20 set out to prevent close packing of the molecules by simultaneously introducing the more bulky tert-butyl group at the C2 position of the anthracene moiety and naphthyl units into the DPA backbone as side groups (emitter 1, for solution) (see Fig. 1). The nondoped emitter 1-based OLED exhibited a saturated deep-blue emission at an EL maximum peak of 444 nm with an International Commission on Illumination (CIE) of (0.15, 0.09). This device exhibited a maximum EQE of 5.17% at with a luminance of . The high color purity and excellent EQE confirmed that the noncoplanar molecular structure of emitter 1 can effectively prevent intermolecular aggregations in the solid film state. Wu et al.26 have also reported that the sterically bulky aryl end-capping group, phenyl-substituted fluorene, provides an effective handle for DPA to prevent close molecular packing. Furthermore, due to these bulky end groups leading to weak intermolecular interactions, emitter 2 exhibited excellent PLQYs both in the solution and film states ( and 0.84 for solution and film, respectively) without excimer formation (see Fig. 1). Accordingly, the emitter 2-based nondoped OLEDs had an EQE value reaching as high as 5.1% () at with a CIE of (0.15, 0.12), which is very close to the National Television System Committee (NTSC) pure blue color. In contrast to the symmetric DPA structure of emitters 1 and 2, Tao et al.33 developed an asymmetric bulky DPA derivative of emitter 3 ( for solution) by introducing different bulky end groups, i.e., pyrene and triphenylamine moieties at the 9- and 10- positions of the anthracene core (see Fig. 1). The end groups in emitter 3 were expected to provide an additional functionality, i.e., efficient hole transportation, because the triphenylamine enables improvement of the hole injection and carrier transport properties. An emitter 3-based device showed highly efficient sky-blue emission [CIE of (0.15, 0.30)] with a maximum efficiency of () in a nondoped device structure. In this device, the turn-on voltage (2.9 V) was also very low because the low energy barrier between hole transporting layer (HTL) and emitter 3 () facilitates efficient hole injection from HTL. More interestingly, even without the layer of HTL, the emitter 3-based nondoped device still had a high maximum LE of with a CIE of (0.15, 0.28). Kim et al.14 developed an emitter 4 that includes a xylene unit (see Fig. 1). A nondoped device based on emitter 4 showed an EQE of 4.37% and a CIE of (0.16, 0.05). FluoreneFluorene derivatives including spiro-type fluorenes have also been proven to be attractive building blocks for blue light-emitting fluorescent materials due to features such as excellent PL properties and good thermal and morphological stability, as well as the ability to tune their optical properties by simple structural modifications. However, the simple fluorene derivatives are susceptible to producing undesirable green emissions with PL quenching, which are attributed to excimer emission arising from aggregates in the film state or the generation of fluorene defects from oxidation. In order to address the color purity and PL quenching problems of fluorene light-emitting materials even at high doping concentrations, Lee et al.29 have reported newly designed fluorene materials that emit deep-blue light, based on 2-(diphenylamino)fluoren-7-ylvinylarene derivatives (emitters 5 and 6) where the diphenylaminofluorene core is combined with a vinylarene unit to obtain the emission of deep-blue light, and the tert-butyl-based blocking group is end-capped to prevent intermolecular interactions (see Fig. 2). From quantum mechanical calculations, it was revealed that the structures of emitters 5 ( for solution) and 6 ( for solution) are nonplanar. The calculated dihedral angles between fluorene (spirofluorene) and the end-capped substituents were found to be 37.9 and 37.4 deg, respectively. The OLED devices based on emitters 5 and 6 with 2-methyl-9,10-di(2-naphthyl)anthracene (MADN) as a host material showed a highly efficient sky-blue emission [CIE of (0.15, 0.17) and 0.20 for emitters 5 and 6, respectively] with EQEs of 6.86 and 7.72% at , respectively. Interestingly, it was also found that the introduction of bulky silicon moieties into the backbone of 2-(diphenylamino)fluoren-7-ylvinylarene as an end-capping unit provides an additional functionality for highly efficient and deep-blue OLED devices,30 i.e., these bulky silicon groups play an important role not only in minimizing the intermolecular interactions, but also in forming a thermally stable and pin-hole-free amorphous thin film through steric and electronic effects (emitters 7 to 11). In optimized OLEDs using emitters 7 to 11 (PLQY , , , , and for film, respectively) as dopants, owing to the steric effect, the silicon end-capping groups efficiently prevent molecular aggregation even at high doping concentrations, and thereby minimize a serious self-quenching effect of the emitting materials in the devices (see Fig. 3). Accordingly, these devices exhibited excellent luminescence of 15,170 to and EQEs of 5.76 to 7.35% with a CIE of (0.15, 0.19) to (0.15, 0.25). In addition, it was revealed that the devices using the emitting material with the triphenylsilyl end-capping groups show better long-term stability than do the devices with the trimethylsilyl end-capping group. Jeon et al.28 designed a different series of spirofluorene derivatives bearing bulky arylamine groups as steric end-capping groups with the expectation of effectively preventing intermolecular interactions. The optimized geometry obtained from density functional theory calculations indicated that the fluorene and benzofluorene groups in the emitters 12 to 15 (PLQY , , , and for solution, respectively) are significantly twisted relative to each other because of the spiro center, resulting in a noncoplanar structure which effectively prevents intermolecular interactions and thereby suppresses molecular recrystallization and aggregation quenching (see Fig. 4). As a result, the OLED devices based on these emitting materials and using an MADN as a host material exhibited astonishingly high maximum EQEs in the range of 6.54 to 8.16% with blue EL emissions (EL of 462 to 465 nm). Aiming to develop highly efficient deep-blue fluorescent fluorene derivatives with a high color purity for nondoped solution-processed OLEDs, Zou et al.17 designed several series of star-shaped oligofluorenes with a propeller-like core, the emitters 16 to 18 (PLQY and 0.74, and 0.81, and 0.84 for solution and film, respectively) (see Fig. 5). The authors emphasized that grafting six fluorene moieties to a benzene core not only constructs a star-shaped architecture, but also generates a highly twisted molecular structure, which contributes to highly efficient emissions of pure blue light. The good solubility and good film-forming capability of these materials enabled their use in the fabrication of solution-processable highly efficient deep-blue OLEDs. The simple double-layer OLEDs based on emitters 16 to 18 displayed a deep-blue emission with a CIE of to 0.17, to 0.08, and exhibited excellent maximum EQEs of up to 6.8%. PyreneDespite emitting very attractive deep-blue light in solution, it is difficult to directly utilize pyrene and its analogues as light-emitting materials for highly efficient blue OLEDs because they have a great propensity to aggregate and to form excimers in the film state, leading to a substantial redshift of the fluorescence emission and a decrease of the efficiency. Therefore, it is desirable to suppress the strong tendency of pyrene moieties to participate in facial stacking interactions. In an attempt to address this issue, Wu et al.23 designed a dipyrenyl benzene derivative, emitter 19 ( and 0.75 for solution and film, respectively), which adopts a twisted geometry with inter-ring torsion angles of 63.2 deg in the crystal state (see Fig. 6). The twisted structure is responsible for the low degree of aggregation and crystallization in the solid state, leading to its favorable light-emission properties in the solid state with good film-forming capabilities. The device reached a maximum efficiency of 5.2% and a very high luminescence of with a CIE of (0.15, 0.11). Chan et al.32 explored a new type of pyrene derivative, pyrene-functionalized calyx[4]arene (emitter 20), where pyrene units are attached to the three-dimensional (3-D) 1,3-alt-calix[4]arene scaffold. The 3-D scaffold played a pivotal role in forcing intramolecular pyrene units to be arranged radially orthogonal to each other, thus ensuring the complete suppression of pyrene aggregation in the solid state (see Fig. 6). The device provided an excellent EQE of 6.4% at with a CIE of (0.15, 0.24). The remarkable EL efficiency of emitter 20 was attributed to the unique ability of the 1,3-alt-calix[4]arene scaffold to promote a variety of rotameric conformations of the pyrene moieties about its single bonds with the scaffold, hence preventing the pyrene units from forming intermolecular stacking interactions and, in turn, limiting concentration-induced quenching of the excited state. Kotchapradist et al.27 developed emitter 21 (see Fig. 6). A nondoped device based on emitter 21 showed an EL maximum wavelength of 436 nm, LE of , and CIE of (0.16, 0.14). Other examplesWei et al.24 have demonstrated that the binaphthyl-type PAH is also a versatile molecular backbone to generate a high-performance blue EL emission. In emitters 22 ( for solution) and 23 ( for solution), the binaphthyl core units are end-capped with arylamine groups (see Fig. 7). The bulky binaphthyl units made these light-emitting materials adopt a noncoplanar configuration, which decreases the tendency to crystallize and weakens the intermolecular interactions in the solid state, hence leading to their pronounced morphological stability (amorphism) and high efficiency. The OLED with emitter 22 doped in emitter 23 as the EML exhibited a pure and deep-blue EL emission, at a wavelength of 444 nm, with CIE coordinates of , , and its EQE reached 8.6% at . Yuan et al.15 developed emitter 24 that includes a bulky PAH. A nondoped device based on emitter 24 showed an LE of 2.10, power efficiency (PE) of 1.88, EQE of 5.02, and CIE of (0.16, 0.05). In order to realize highly efficient nondoped blue OLEDs having both high EQEs and high PEs by effectively improving the injection of charge carriers into the light-emitting layers from the adjacent layers, Kuo et al.22 employed a specific -type imidazole derivative, a phenanthroimidazolyl (PPI) moiety, as an OLED-emitting material. By using this building block, they developed a new series of blue light-emitting materials, emitters 25 to 27 (PLQY , , and for solution, respectively), in which the phenyl substituents attached to the PPI groups are highly twisted around the PPI groups with dihedral angles of , while the inter-ring torsion angles between the biphenyl group and two PPI groups are in the single-crystal state (see Fig. 7). The nondoped OLEDs based on these emitting materials gave very high EQEs (5.26 to 6.31%) and pure blue emission with a CIE of (0.15, 0.09) to (0.15, 0.15). In particular, among these devices, the emitter 25-based device resulted in a very low turn-on voltage (3.0 V) and a high PE of . Chen et al.16 developed an emitter 28 using staggered face-to-face molecular stacking as a means to design a deep-blue EL material (see Fig. 7). A nondoped device based on emitter 28 showed an LE of 2.06, PE of 1.60, EQE of 4.93, and CIE of (0.16, 0.05). Taking advantage of the characteristic optical properties of indenopyrazine units, such as their highly pure blue colors with a narrow full width half maximum (FWHM), Park et al.18 developed a new indenopyrazine derivative, emitter 29 ( for film), with bulky aryl side groups (see Fig. 7). Emitter 29 showed a distinctive blue emission (at a wavelength of 444 nm) originating from the indenopyrazine core in the film state. The authors pointed out that the introduction of an electron-accepting imine group into the core of the -conjugated backbone stabilizes and reduces the LUMO level and, thus, drastically facilitates the electron injection and electron transport capabilities. Nondoped OLED devices based on emitter 29 exhibited a blue EL emission at 453 nm with a narrow FWHM (47 nm) [CIE of (0.15, 0.07)] and highly efficient EQEs of 5.1%. 2.1.2.Donor-/acceptor-type emittersThe incorporation of electron-donor (D) and electron-acceptor (A) moieties into light-emitting materials has been considered one of the promising molecular design strategies for enhancing EQEs of OLEDs because it provides the light-emitting materials while facilitating exciton formation (via stable radical cations and anions) and improves the charge balance in the light-emitting layers of OLEDs. However, the D-A bipolar system tends to produce charge-transfer (CT) features from the donor unit to the acceptor unit that could cause a large bathochromic effect, thus impairing the color purity of the blue emission. Because of the intrinsic wide band gap character of the blue light-emitting materials, it is a great challenge to develop highly efficient blue light-emitting materials for OLEDs by utilizing D-A bipolar systems. Lin et al.31 successfully explored a new D-A bipolar fluorescent chromophore, emitter 30 ( and 0.51 for solution and film, respectively), for highly efficient blue OLEDs by combining a carbazole donor and a dimesitylborane acceptor into one -conjugation (electron-rich thiophene ring as a bridge unit) system (see Fig. 8). The nondoped OLEDs based on emitter 30 showed a blue EL emission [, CIE of (0.13, 0.21)] without any exciplex formation between this emitter and HTL material, and showed a very high maximum EQE of 6.9%. It was pointed out that all three essential units in emitter 30, i.e., carbazole, dimesitylborane, and thiophene, play important roles in obtaining highly efficient blue OLEDs. Li et al.25 have reported an effective way to construct highly efficient blue OLEDs by using a twisting D-A molecule, emitter 31 ( and 0.90 for solution and film, respectively), consisting of two main components: triphenylamine as the donor moiety and 1,2-diphenyl-1H-phenanthro[9,10-d]imidazole (PPI) as the acceptor (see Fig. 8). In spite of these molecules being CT type molecules, emitter 31 very rarely showed a deep-blue emission ( of ca. 440 nm) with very high PLQYs of 90% in both the solution and film states. In addition, a nondoped OLED with emitter 31 displayed not only an excellent deep-blue EL performance [CIE of (0.15, 0.11)], but also a maximum EQE of 5.02%. Jeong et al.19 developed a deep-blue emitter 32 using a derivative that connected triphenylamine and benzimidazole (see Fig. 8). A nondoped device based on emitter 32 showed an EL maximum wavelength of 452 nm, EQE of 4.67%, and CIE of (0.15, 0.08). 2.1.3.Organometallic-type emittersAlthough a variety of phosphorescent metal complexes have been developed for high-performance OLEDs, few fluorescent metal complexes, especially those that emit blue light, have been developed and investigated so far. Recently, Peng et al.21 reported that a simple structural modification of a blue light-emitting organometallic-type material can dramatically improve its electroluminescent properties. They introduced a methyl unit onto the pyridyl ring of the ligand in the original beryllium complex, bis(2-(2-hydronyphenyl)-pyridine)beryllium (Bepp2), and thus prepared a novel sterically hindered beryllium complex, emitter 33 ( for solution), for highly efficient nondoped blue OLEDs (see Fig. 8). In the nondoped double-layered OLED device based on emitter 33, a high quality of blue light emission [CIE of (0.14, 0.09)] was achieved because the introduced methyl unit effectively prevented exciplex formation with a hole-transporting material. The EQE of this device surprisingly reached 5.4% at . 2.2.Red Fluorescent EmittersOwing to the inherently low PLQYs from the red fluorescent chromophores, the red light-emitting fluorescent materials exceeding EQEs of 5% have rarely been found in recent official papers. Although it was not an academic paper, a report by Idemitsu Kosan Co., Ltd. in 2007 announced red fluorescence with a high efficiency of and a device lifetime of 160,000 h. This report, however, will not be discussed further in this review since a specific chemical structure was not provided.35 Nevertheless, Yang et al.36 reported a high-performance near-IR OLED based on a donor-acceptor-donor conjugated oligomer, emitter 34, that has a narrow band gap (ca. 1.9 eV) by combining the strong donor dihydrothieno dioxin with the strong acceptor thiadiazolo quinoxaline (see Fig. 9). Emitter 34 exhibited a PL maximum at 698 nm and its PLQY was measured to be 21% in solution. The emitter 34-based OLED with a 3.5 wt% doping concentration in the EML showed a maximum EQE of 1.6%. Notably, however, the EQE of the device was further enhanced almost twofold () by incorporating a phosphorescent sensitizer in the EML together with the host and the emitter, making a so-called sensitized fluorescent OLED device architecture, which harvests both singlet and triplet excitons and transfers them to emitter 34 through a Förster-type energy transfer process. Unfortunately, a maximum EQE of only 1.6% was obtained for the device with a 5 wt% doping concentration of emitter 34 because of the inevitable aggregation-induced fluorescence quenching. Studies on TADF, recently presented by the Adachi group, revealed high-efficiency red fluorescent materials. As a representative case, 4,4′,4′′-(-heptaazaphenalene-2,5,8-triyl)tris(N,N-bis(4-(tert-butyl)phenyl)aniline) is a TADF material studied based on the heptazine structure that showed a TADF effect with a small energy difference between S1 and T1 () of 0.27 eV. It exhibited a high EL efficiency of and EQE of 17.5%.37 In addition, as a result of synthesizing 2,6-bis[4-(diphenylamino)phenyl]anthraquinone and applying it as a dopant in a device, a maximum EL of 624 nm and CIE of (0.16, 0.39) were achieved, along with an extremely high EQE of 12.5%. However, the results for the lifetime of this material have not yet been reported.34 3.Phosphorescent Materials for Two-Color White OLEDsPhosphorescent materials can theoretically achieve a 100% internal quantum efficiency as they can harvest both singlet and triplet states. In general, triplet exciton harvesting through intersystem crossing occurs at a phosphorescent molecule containing a heavy atom that induces strong spin-orbit coupling. Many studies have been conducted on transition metal complexes using iridium, platinum, and osmium. Moreover, since phosphorescent materials that include heavy atoms cannot be used by themselves in devices, the usage of host materials is essential although some phosphorescence materials were applied to an OLED device as nondoped emitters.38 This section of the article reviews not only dopant materials used for emission, but also host materials used for the generation of phosphorescence. 3.1.Phosphorescent Blue Host for White OLEDsBlue phosphorescent host materials are required to achieve highly efficient lighting with white OLEDs, and many studies of such materials are underway. Since the triplet energy of blue phosphorescent dopant materials is, in general, 2.70 to 2.75 eV, the host material should have a higher triplet energy. When the triplet energy of the host material is lower than that of the dopant material, back energy transfer can occur from the dopant to the host. Such back energy transfer can have disadvantageous consequences such as triplet exciton quenching and low device efficiency. Therefore, blue phosphorescent host materials must have a triplet energy of 2.75 eV or higher in order to prevent the back energy transfer of excitons. Representative core structures with high triplet energy include carbazole, arylsilane, dibenzofuran, and dibenzothiophene. For the development of blue phosphorescent host materials with a high triplet energy, it is necessary to reduce the conjugation length to achieve a wide band gap. For example, the introduction of carbon, silicon, and phosphine oxide units can reduce the conjugation length within the molecule. Introducing a sterically hindered structure can also decrease the conjugation length. As mentioned earlier, since increasing the triplet energy is extremely important for improving device performance in blue phosphorescent host materials, we are going to focus on reviewing phosphorescent host materials with regard to their core structures and triplet energy levels. Since the carbazole core has a high triplet energy of 3.02 eV and good hole transport properties, it can be used as a blue phosphorescent host material.39 Substituents on the carbazole core could affect the triplet energy of the phosphorescent host material. Emitter 35, a biphenyl para-linked to carbazoles, is most commonly used as a phosphorescent host material (see Fig. 10). However, in the case of emitter 35, the triplet energy was slightly smaller at 2.6 eV than the 2.7 eV value for emitter 70 when used as the dopant (see Fig. 11). It is hard to achieve a high efficiency with this system due to the back energy transfer from emitter 70 to emitter 35.40 In the case of emitter 36, a derivative of carbazole in which two carbazole units are linked to the meta positions of a phenyl group, a high triplet energy of 2.9 eV,41 and a wide band gap is shown for the blue phosphorescent host material (see Fig. 10). Emitter 37,42 in which two carbazoles are linked to the 4 and 4′ positions of a 2,2′-dimethylbiphenyl unit, has a chemical structure similar to that of emitter 35. The triplet energy (3.0 eV) of emitter 37 was shown to be higher than that of emitter 35 (see Fig. 10). Emitter 37 maintained the triplet energy of carbazole because the introduction of the methyl groups on the biphenyl unit distorted the chemical structure. Other phosphorescent host materials based on carbazole core compounds, such as emitters 38,43 39,44 40,45 41,46 43,47 and 44,47 are summarized in Table 2 (see Fig. 10) Table 2Energy levels of blue phosphorescent hosts.
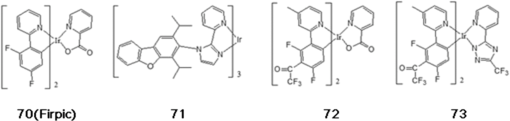
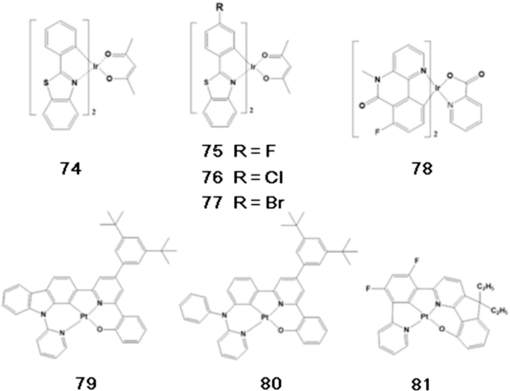
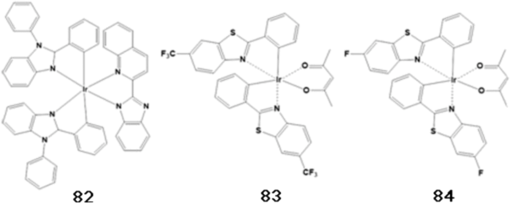
Note: HOMO, highest occupied molecular orbital; LUMO, lowest unoccupied molecular orbital; ET, triplet energy level; Tg glass transition temperature. Substituting a triphenylsilyl unit for a hydrogen in the carbazole core improved the thermal stability without decreasing the triplet energy of the material. While the glass transition temperature of emitter 36 was shown to be 55°C, that of emitter 45, in which a triphenylsilyl group is bonded to an emitter 36 unit, increased to 101°C (see Fig. 10).51 Emitter 46, in which a triphenylsilyl group is bonded to two emitter 36 units, also showed a high glass transition temperature of 148°C (see Fig. 10).52 The triplet energy of emitter 45 was shown to be 2.90 eV and the triplet energy of emitter 46 was shown to be 3.01 eV. Emitters 47,53 48,50 and 4954 were also developed by adapting phenyl carbazole units with triphenylsilyl or diphenylsilane groups (see Fig. 10). These materials showed a high triplet energy and a high glass transition temperature. Host materials with a high triplet energy and good charge transport characteristics were developed by including moieties such as dibenzothiophene and dibenzofuran core groups to phosphine oxide units. Phosphine oxide units can enhance electron transport properties without decreasing the triplet energy of the core structure. Emitter 50,55 in which two diphenylphosphine oxide units are bonded to the 3 and 6 positions of phenyl carbazole, showed a high triplet energy of 3.02 eV and balanced charge transport properties (see Fig. 10). Emitter 51,56 which substituted diphenylphosphine oxide units in the 2 and 7 positions of phenyl carbazole, showed a lower triplet energy and band gap compared to emitter 50 (see Fig. 10). Emitter 52,57 which substituted a diphenylphosphine oxide unit into emitter 36, was shown to have a triplet energy similar to that of emitter 36, but with improved electron transport properties (see Fig. 10). Host materials with a high triplet energy and bipolar charge transport ability were developed by substituting carbazole and diphenylphosphine oxide moieties into a dibenzothiophene or dibenzofuran core unit. High triplet energy host materials of emitters 53, 54, and 5558 were synthesized, and their charge transport properties were compared (see Fig. 10). Emitter 54, having one carbazole unit and one diphenylphosphine oxide unit bonded to the dibenzothiophene core, showed bipolar charge transport properties (see Fig. 10). Dibenzofuran derivatives of emitters 56, 57, and 5859 were studied by fabricating devices with single or mixed host structures (see Fig. 10). Host materials that use an Si atom to disconnect the conjugation of phenyl units could increase the band gap and triplet energy. Host materials that use an Si atom to disconnect the conjugation of phenyl units could increase the band gap and triplet energy. Emitters 59,48,49 60,48,49 and 6149 were developed by substitutions at different positions and showed high triplet energy values (see Fig. 10). But the deep HOMO level of emitters 59, 60, and 61 limits hole injection from the HTL to the EML and causes an increase of the driving voltage (see Fig. 10). The additional phosphorescent blue host materials were also summarized in Table 2. 3.2.Blue Phosphorescent DopantsEmitter 70 (known as Firpic) has been widely used as a blue phosphorescent dopant (see Fig. 11). However, many efforts have been made to improve this dopant since it actually emits more in the greenish blue wavelength range and has a short device lifetime. Zhuang et al.68 developed emitter 71, a homoleptic triscyclometalated iridium(III) complex in which the ligand is substituted as one kind, to compare its EL performance with that of emitter 70 (see Fig. 11). While a device that used emitter 70 was shown to have an LE of , PE of , and EQE of 13.5%, a device that used emitter 71 showed an improved efficiency, with an LE of , PE of , and EQE of 23.1%. In addition, the lifetime of the device that used emitter 71 was 5.8 h, which is considerably longer than the 0.1-h lifetime of the emitter 70 device. Nonetheless, the device that used emitter 71 had a CIE of (0.19, 0.44), which is more greenish blue compared to the (0.16, 0.36) CIE of the device that used emitter 70. Lee et al.69 developed emitters 72 and 73 as perfluoro carbonyl-substituted iridium complexes for the development of phosphorescent blue dopants with high efficiency and emission of deep-blue light (see Fig. 11). The device that used emitter 72 was shown to have an LE of , PE of , and high EQE of 17.1%, as well as a CIE of (0.141, 0.158), showing a deeper blue emission compared to that of emitter 70. Emitter 73 had a CIE of (0.147, 0.116) in the deep-blue region. 3.3.Yellow Light Emitters for OLED LightingIn 2013, Fan et al. developed a new iridium complex.70 The effects of substituting different halogens (F, Cl, Br) in this structure on optoelectronic properties were systematically studied. While the halogen-free emitter 74 showed an LE of and PE of , the Ir complex substituted with halogens showed increased PE values in the range of 55.9 to . In particular, emitter 75, which contains an F atom, had an LE of and PE of with a yellow emission CIE of (0.45, 0.53), showing improved device efficiency compared to halogen-free emitter 74. In 2014, Jou et al. developed a new yellow light-emitting iridium complex that can be prepared by wet and dry processes.71 As shown in Fig. 12, the synthesized material (78) substituting hydrogen in the 2 position with fluorine not only blocked molecular packing, but also decreased self-quenching, and provided a high PLQY of 71%. In the case of the dry process using vapor deposition, high efficiency was found with an EQE of 22.6% and PE of . The highest efficiency shown for a yellow light-emitting device fabricated using the wet spin-coating method was an EQE of 18.5% and PE of . In 2013, Lai et al. synthesized a new derivative of a platinum(II) complex, which included a dianionic tetradentate ligand (see Fig. 12).72 Among the synthesized materials, emitter 79 showed a high efficiency with a quantum yield of 86% in the solution state. When prepared into a device, a PE of was shown with an emission of yellow light at 568 nm and a CIE of (0.52, 0.47). When emitter 57 was used as a blue light-emitting dopant and applied to two-color white OLEDs, a high efficiency was reported with a CIE of (0.34, 0.44) and PE of .73 Later on, in 2014, emitters 80 and 81, shown in Fig. 12, were synthesized by modifying the Pt(II) complex presented above. In the case of emitter 80, an extremely high efficiency of yellow light emission was shown with a PE of and EQE of 26%. Emitter 81 was applied to a white OLED as a single emitter and showed a CIE of (0.38, 0.47) and EQE of 25.1%. 3.4.Orange Light Emitters for OLED LightingIn 2013, Cao et al. synthesized emitter 82, which emits orange light at a wavelength of 573 nm (see Fig. 13).74 The synthesized material showed high efficiency with an LE of and PE of for a CIE of (0.53, 0.46). An LE of and PE of were shown when emitter 82 was used as a dopant in a white OLED device whose structure consisted of indium tin oxide (ITO)/MoO3 (3 nm)/4,4′-cyclohexylidenebis[N,N-bis(4-methylphenyl)benzenamine] (TAPC) (30 nm)/tris(4-carbazoyl-9-ylphenyl)amine (TCTA) (5 nm)/2,7-bis(diphenylphosphoryl)-9-(4-diphenylamino)phenyl-9’-phenyl-fluorene (POAPF):emitter 82 (7%) (6 nm)/POAPF:FIr6 (10%) (24 nm)/BmPyPhB (35 nm)/Liq (1 nm)/Mg:Ag. A color rendering index (CRI) of 80 was also shown and the CIE was (0.33, 0.36), which is close to the standard white emission. In 2011, Wang et al. developed an orange light-emitting iridium complex with a new 2-phenylbenzothiazole ligand.75 This emitter 83 showed extremely high efficiency for a CIE of (0.52, 0.47), with an LE of and PE of (see Fig. 13). Moreover, a two-color white OLED device fabricated with the structure ITO/poly(3,4-ethylenedioxythiophene) polystyrene sulfonate (PEDOT:PSS) (40 nm)/orange phosphor:emitter 35 (1 wt%, 10 nm)/ emitter 70:emitter 35 (10 wt%, 20 nm)/1,3,5-tris(1-phenyl-1H-benzimidazol-2-yl)benzene (TPBI) (45 nm)/LiF (1 nm)/Al, using emitter 70 as the source of blue light showed a CIE of (0.35, 0.44), LE of , and high EQE of 26.2%. 3.5.Red Light Emitters for OLED LightingIn 2012, Wang et al. synthesized a new iridium complex that used 2-phenylbenzothiozole bonded with the -phenylnaphthyl group as a ligand (emitters 85 and 86 in Fig. 14).76 As a result of fabricating devices using emitter 35 as the host and emitters 85 and 86 as dopants, emitters 85 and 86 doped 5% in the device showed CIEs of (0.56, 0.44) and (0.60, 0.40), respectively, and exhibited high-efficiency emission of red light with PEs of 6.56 and , respectively, and EQEs of 6.49 and 8.73%, respectively. In addition, as a result of preparing a white OLED device with an ITO/PEDOT:PSS (40 nm)/emitter 86 (0.5 wt%):emitter 70 (8 wt%):emitter 35 (60 nm)/TPBI (45 nm)/LiF (1 nm)/Al structure, a PE of and an EQE of 4.9% were shown, as well as a near-real white emission with a CIE of (0.33, 0.35). In 2012, Tavasli et al. synthesized many new Ir complexes based on the carbazolyl pyridine ligand (see Fig. 14).77 Among the synthesized materials, emitters 87 and 88 showed emission of red light with values of 637 and 633 nm, respectively. As a result of applying these materials in a simple device consisting of ITO/PEDOT:PSS-HIL 1.5 (60 nm)/poly(9-vinylcarbazole) (PVK):2-(4-biphenylyl)-5-(4-tert-butylphenyl)-1,3,4-oxadiazole (PBD):Ir complex (90 nm)/Ba (4 nm)/Al (100 nm), made using the solution spin-coating method, a high efficiency was shown with a PE of 0.6 to and EQE of 4 to 5.8%. Many existing phosphorescent dopants that emit red light with high efficiency use Ir complexes such as (tris[1-phenylisoquinolinato-C2,N]iridium(III)).78 However, several new red phosphorescent dopants have recently been developed using other heavy metals. In 2012, Du et al. developed emitter 89 with a new 3-(thiazol-2-yl), 3-(benzothiazol-2-yl), 3-(imidazol-2-yl), and 3-(benzimidazol-2-yl) azole chelate.79 Os(II) complexes exhibit emissions of green to red light depending on the substituted group as shown in Fig. 15. In particular, emitter 89 was applied to a device made up of ITO/PEDOT:PSS/VB-FNPD/TCTA:emitter 89/TPBI/CsF/Al that emits red light with a CIE of (0.63, 0.37), yielding an LE of , a high PE of , and an excellent EQE of 15.6%. In 2012, Fukagawa et al. synthesized new platinum complexes, shown in Figure 15, that emit red light.80 In a device made up of ITO/ND-1501 (30 nm)/N,N’-di(1-naphthyl)-N,N’-diphenylbenzidine (-NPD) (50 nm)/ (90 or 91 6 wt% doped, 35 nm)/ETM-143 (40 nm)/LiF (1 nm)/Al (100 nm), emitters 90 and 91 were shown to emit red light with a CIE of (0.66, 0.33) and (0.65,0.34), respectively. These emitters were shown to have PEs of 20.7 and and maximum EQEs of 18.5 and 18.2%, and hence to be more efficient than existing Ir complexes. They also displayed high device stability with lifetimes of 8200 and 15,000 h at . 4.Latest Trend of Industrial Research on White OLEDsIn addition to the materials described in academic papers, diverse and excellent lighting device performances based on undisclosed materials developed in industry have also been proposed. Major outcomes of industrial research that were presented in 2013 are as follows. LG Chem. showed a PE of for a three-stack tandem structure and reported a lifetime (i.e., LT70) of 26,000 h at .81 Universal Display Corp. reported a PE of and lifetime (LT70) of 27,500 h at using phosphorescent stacked OLEDs.82 Hitachi reported a PE of for a highly efficient white OLED device made up of a single EML that includes three types of dopants.83 In addition, Panasonic Corp. reported a high PE of at and at for all of their phosphorescent white OLEDs.84 Major outcomes of industrial research that were presented in 2014 are as follows. Universal Display Corp. applied a phosphorescent material emitting R, G, B colors and reported a color-tunable white OLED lighting panel with high efficiency. This material showed a high efficiency of and a correlated color temperature of 2700 K at without using outcoupling enhancement. Using 1.53-fold outcoupling enhancement yielded a high efficiency of at .85 Konica-Minolta used an optimization method using angular light distribution of organic layers and light extraction structure based on a high refractive index substrate to prepare white OLEDs showing high efficacy of at .86 ETRI applied a random nanostructured scattering layer and reported a white OLED showing and CRI of 80.87 SEL combined exciplex-triplet energy transfer and triplet-triplet annihilation to develop a single-unit hybrid OLED and announced a PE of and lifetime (LT70) of 180,000 h at .88 According to noticeable press releases based on such results, LG Chem. reported in 2014 that it had successfully developed OLED light panel products with an efficiency of and long lifetime of 40,000 h (see Fig. 16). These products can compete against fluorescent sources of light (, 20,000 h) as well as LEDs ( or higher, 50,000 h). In addition, this performance is very high compared to the average efficiency of OLED panel products by other lighting companies, which is still . According to UBI Research, the OLED light panel market is expected to start flourishing in 2015 and expand to markets of million USD($) in 2016 and 4.8 billion USD($) in 2020, showing an explosive growth of per year. Research and development in this field will become increasingly active.89 5.ConclusionsWhite OLED research is garnering a lot of attention in industrial and academic settings. There is considerable demand in the lighting field to develop and commercialize next-generation products with high efficiency and lifetime to replace fluorescent and incandescent lights, and many research groups today are striving to embody OLED lighting with these advantageous features. In order to manufacture high-efficiency and long-lifetime white OLEDs, it is most important to have a systematic and strategic understanding of the development of relevant materials. In this review article, fluorescent and phosphorescent materials that are being applied to white OLEDs have been categorized, and the chemical structures and device performances of the important blue, orange, and red light-emitting materials have been summarized. Such a systematic classification and understanding of the materials that have already been reported on can aid the development and study of new light-emitting materials through quantitative and qualitative approaches. AcknowledgmentsThis research was supported by a grant from the Technology Development Program for Strategic Core Materials funded by the Ministry of Trade, Industry & Energy, Republic of Korea (Project No. 10047758). This research was supported by a grant from the Fundamental R&D Program for Core Technology of Materials funded by the Ministry of Trade, Industry and Energy, Republic of Korea (Project No. 10050215) ReferencesC. W. Tang and S. A. Vanslyke,
“Organic electroluminescent diodes,”
Appl. Phys. Lett., 51 913
(1987). http://dx.doi.org/10.1063/1.98799 APPLAB 0003-6951 Google Scholar
Z. Shen et al.,
“Three-color, tunable, organic light-emitting devices,”
Science, 276 2009
–2011
(1997). http://dx.doi.org/10.1126/science.276.5321.2009 SCIEAS 0036-8075 Google Scholar
S. R. Forrest,
“The road to high efficiency organic light emitting devices,”
Org. Electron., 4 45
–48
(2003). http://dx.doi.org/10.1016/j.orgel.2003.08.014 1566-1199 Google Scholar
A. R. Duggal et al.,
“Organic light-emitting devices for illumination quality white light,”
Appl. Phys. Lett., 80 3470
(2002). http://dx.doi.org/10.1063/1.1478786 APPLAB 0003-6951 Google Scholar
B. W. D’Andrade and S. R. Forrest,
“White organic light-emitting devices for solid-state lighting,”
Adv. Mater., 16 1585
–1595
(2004). http://dx.doi.org/10.1002/adma.200400684 ADVMEW 0935-9648 Google Scholar
C. P. Wang et al.,
“High-efficiency flexible white organic light-emitting diodes,”
J. Mater. Chem., 20 6626
–6629
(2010). http://dx.doi.org/10.1039/c0jm01348j JMACEP 0959-9428 Google Scholar
Y. L. Chang et al.,
“Highly efficient warm white organic light-emitting diodes by triplet exciton conversion,”
Adv. Funct. Mater., 23 705
–712
(2013). http://dx.doi.org/10.1002/adfm.201201858 AFMDC6 1616-3028 Google Scholar
S. Reineke et al.,
“White organic light-emitting diodes with fluorescent tube efficiency,”
Nature, 459 234
–238
(2009). http://dx.doi.org/10.1038/nature08003 NATUAS 0028-0836 Google Scholar
M. Thomschke et al.,
“Highly efficient white top-emitting organic light-emitting diodes comprising laminated microlens films,”
Nano Lett., 12 424
–428
(2012). http://dx.doi.org/10.1021/nl203743p NALEFD 1530-6984 Google Scholar
X. Yang et al.,
“Modified 4,4′,4′′-Tri(N-carbazolyl)triphenylamine as a versatile bipolar host for highly efficient blue, orange, and white organic light-emitting diodes,”
J. Phys. Chem. C, 116 15041
–15047
(2012). http://dx.doi.org/10.1021/jp3034566 1932-7447 Google Scholar
L. Duan et al.,
“Controlling the recombination zone of white organic light-emitting diodes with extremely long lifetimes,”
Adv. Funct. Mater., 21 3540
–3545
(2011). http://dx.doi.org/10.1002/adfm.201100943 AFMDC6 1616-3028 Google Scholar
S. M. Shen, Y. C. Tsai and J. H. Jou,
“Pure white organic light-emitting diodes with lifetime approaching the longevity of yellow emitter,”
ACS Appl. Mater. Interfaces, 3 3134
–3139
(2011). http://dx.doi.org/10.1021/am200612z AAMICK 1944-8244 Google Scholar
S. Wu et al.,
“High-efficiency deep-blue organic light-emitting diodes based on a thermally activated delayed fluorescence emitter,”
J. Mater. Chem. C, 2 421
–424
(2014). http://dx.doi.org/10.1039/C3TC31936A JMCCCX 2050-7534 Google Scholar
R. Kim et al.,
“Extremely deep blue and highly efficient non-doped organic light emitting diodes using an asymmetric anthracene derivative with a xylene unit,”
Chem. Commun., 49 4664
–4666
(2013). http://dx.doi.org/10.1039/c3cc41441h 1359-7345 Google Scholar
Y. Yuan et al.,
“Bipolar phenanthroimidazole derivatives containing bulky polyaromatic hydrocarbons for nondoped blue electroluminescence devices with high efficiency and low efficiency roll-off,”
Chem. Mater., 25 4957
–4965
(2013). http://dx.doi.org/10.1021/cm4030414 CMATEX 0897-4756 Google Scholar
W. C. Chen et al.,
“Staggered face-to-face molecular stacking as a strategy for designing deep-blue electroluminescent materials with high carrier mobility,”
Adv. Opt. Mater., 2 626
–631
(2014). http://dx.doi.org/10.1002/adom.201400078 AOMDAX 2195-1071 Google Scholar
Y. Zou et al.,
“Unexpected propeller-like hexakis(fluoren-2-yl)benzene cores for six-arm star-shaped oligofluorenes: highly efficient deep-blue fluorescent emitters and good hole-transporting materials,”
Adv. Funct. Mater., 23 1781
–1788
(2013). http://dx.doi.org/10.1002/adfm.201202286 AFMDC6 1616-3028 Google Scholar
Y. Park et al.,
“An aromatic imine group enhances the EL efficiency and carrier transport properties of highly efficient blue emitter for OLEDs,”
J. Mater. Chem., 20 5930
–5936
(2010). http://dx.doi.org/10.1039/c0jm00581a JMACEP 0959-9428 Google Scholar
S. Jeong et al.,
“Efficient deep-blue emitters based on triphenylamine-linked benzimidazole derivatives for nondoped fluorescent organic light-emitting diodes,”
Org. Electron., 14 2497
–2504
(2013). http://dx.doi.org/10.1016/j.orgel.2013.06.022 1566-1199 Google Scholar
C. J. Zheng et al.,
“Highly efficient non-doped deep-blue organic light-emitting diodes based on anthracene derivatives,”
J. Mater. Chem., 20 1560
–1566
(2010). http://dx.doi.org/10.1039/b918739a JMACEP 0959-9428 Google Scholar
T. Peng et al.,
“Novel beryllium complex as the non-doped emitter for highly efficient deep-blue organic light-emitting diode,”
Org. Electron., 12 1914
–1919
(2011). http://dx.doi.org/10.1016/j.orgel.2011.08.006 1566-1199 Google Scholar
C. J. Kuo et al.,
“Bis(phenanthroimidazolyl)bisphenyl derivatives as saturated blue emitters for electroluminescent devices,”
J. Mater. Chem., 19 1865
–1871
(2009). http://dx.doi.org/10.1039/b816327h JMACEP 0959-9428 Google Scholar
K. C. Wu et al.,
“The photophysical properties of dipyrenylbenzenes and their application as exceedingly efficient blue emitters for electroluminescent devices,”
Adv. Funct. Mater., 18 67
–75
(2008). http://dx.doi.org/10.1002/adfm.200700803 AFMDC6 1616-3028 Google Scholar
B. Wei et al.,
“Stable, glassy, and versatile binaphthalene derivatives capable of efficient hole transport, hosting, and deep-blue light emission,”
Adv. Funct. Mater., 20 2448
–2458
(2010). http://dx.doi.org/10.1002/adfm.201000299 AFMDC6 1616-3028 Google Scholar
W. Li et al.,
“A twisting donor-acceptor molecule with an intercrossed excited state for highly efficient, deep-blue electroluminescence,”
Adv. Funct. Mater., 22 2797
–2803
(2012). http://dx.doi.org/10.1002/adfm.201200116 AFMDC6 1616-3028 Google Scholar
C. H. Wu et al.,
“Efficient non-doped blue light-emitting diodes incorporating an anthracene derivative end-capped with fluorene groups,”
J. Mater. Chem., 19 1464
–1470
(2009). http://dx.doi.org/10.1039/b817031b JMACEP 0959-9428 Google Scholar
P. Kotchapradist et al.,
“Pyrene-functionalized carbazole derivatives as non-doped blue emitters for highly efficient blue organic emitting diodes,”
J. Mater. Chem. C, 1 4916
–4924
(2013). http://dx.doi.org/10.1039/c3tc30719k JMCCCX 2050-7534 Google Scholar
Y. M. Jeon et al.,
“Deep-blue OLEDs using novel efficient spiro-type dopant materials,”
Org. Electron., 11 1844
–1852
(2010). http://dx.doi.org/10.1016/j.orgel.2010.08.007 1566-1199 Google Scholar
K. H. Lee et al.,
“Highly efficient blue organic light-emitting diodes based on 2-(diphenylamino)fluoren-7-ylvinylarene derivatives that bear a tert-butyl group,”
Chem. Eur. J., 17 12994
–13006
(2011). http://dx.doi.org/10.1002/chem.v17.46 CEUJED 0947-6539 Google Scholar
K. H. Lee et al.,
“Molecular engineering of blue fluorescent molecules based on silicon end-capped diphenylaminofluorene derivatives for efficient organic light-emitting materials,”
Adv. Funct. Mater., 20 1345
–1358
(2010). http://dx.doi.org/10.1002/adfm.200901895 AFMDC6 1616-3028 Google Scholar
S. L. Lin et al.,
“Highly efficient carbazole--dimesitylborane bipolar fluorophores for nondoped blue organic light-emitting diodes,”
Adv. Mater., 20 3947
–3952
(2008). http://dx.doi.org/10.1002/adma.200801023 ADVMEW 0935-9648 Google Scholar
K. L. Chan et al.,
“High-efficiency pyrene-based blue light emitting diodes: aggregation suppression using a calixarene 3D-scaffold,”
Chem. Commun., 48 5106
–5108
(2012). http://dx.doi.org/10.1039/c2cc30995e 1359-7345 Google Scholar
S. Tao et al.,
“Highly efficient nondoped blue organic light-emitting diodes based on anthracene-triphenylamine derivatives,”
J. Phys. Chem. C, 112 14603
–14606
(2008). http://dx.doi.org/10.1021/jp803957p 1932-7447 Google Scholar
Q. Zhang et al.,
“Anthraquinone-based intramolecular charge-transfer compounds: computational molecular design, thermally activated delayed fluorescence, and highly efficient red electroluminescence,”
J. Am. Chem. Soc., 136 18070
–18081
(2014). http://dx.doi.org/10.1021/ja510144h JACSAT 0002-7863 Google Scholar
H. Kuma et al.,
“Highly efficient white OLEDs using RGB fluorescent materials,”
Proc. SID, 38 1504
–1507
(2007). http://dx.doi.org/10.1889/1.2785601 SIDPAA 0734-1768 Google Scholar
Y. Yang et al.,
“Efficient near-infrared organic light-emitting devices based on low-gap fluorescent oligomers,”
J. Appl. Phys., 106 044509
(2009). http://dx.doi.org/10.1063/1.3204947 JAPIAU 0021-8979 Google Scholar
J. Li et al.,
“Highly efficient organic light-emitting diode based on a hidden thermally activated delayed fluorescence channel in a heptazine derivative,”
Adv. Mater., 25 3319
–3323
(2013). http://dx.doi.org/10.1002/adma.201300575 ADVMEW 0935-9648 Google Scholar
Y. H. Song et al.,
“Bright and efficient, non-doped, phosphorescent organic red-light-emitting diodes,”
Adv. Funct. Mater., 14 1221
–1226
(2004). http://dx.doi.org/10.1002/adfm.200400137 AFMDC6 1616-3028 Google Scholar
J. B. Birks, Photophysics of Aromatic Compounds, Wiley, New York
(1970). Google Scholar
C. Adachi et al.,
“Nearly 100% internal phosphorescence efficiency in an organic light-emitting device,”
J. Appl. Phys., 90 5048
(2001). http://dx.doi.org/10.1063/1.1409582 JAPIAU 0021-8979 Google Scholar
R. J. Holmes et al.,
“Blue organic electrophosphorescence using exothermic host-guest energy transfer,”
Appl. Phys. Lett., 82 2422
(2003). http://dx.doi.org/10.1063/1.1568146 APPLAB 0003-6951 Google Scholar
S. Tokito et al.,
“Confinement of triplet energy on phosphorescent molecules for highly efficient organic blue-light emitting devices,”
Appl. Phys. Lett., 83 569
(2003). http://dx.doi.org/10.1063/1.1594834 APPLAB 0003-6951 Google Scholar
G. T. Lei et al.,
“Highly efficient blue electrophosphorescent devices with a novel host material,”
Synth. Met., 144 249
–252
(2004). http://dx.doi.org/10.1016/j.synthmet.2004.03.010 SYMEDZ 0379-6779 Google Scholar
D. R. Whang et al.,
“A highly efficient wide-band-gap host material for blue electrophosphorescent light-emitting devices,”
Appl. Phys. Lett., 91 233501
(2007). http://dx.doi.org/10.1063/1.2821116 APPLAB 0003-6951 Google Scholar
H. Fukagawa et al.,
“Highly efficient, deep-blue phosphorescent organic light emitting diodes with a double-emitting layer structure,”
Appl. Phys. Lett., 93 133312
(2008). http://dx.doi.org/10.1063/1.2996572 APPLAB 0003-6951 Google Scholar
Y. Agata, H. Shimizu and J. Kido,
“Syntheses and properties of novel quarterphenylene-based materials for blue organic light-emitting devices,”
Chem. Lett., 36 316
–317
(2007). http://dx.doi.org/10.1246/cl.2007.316 CMLTAG 0366-7022 Google Scholar
L. S. Cui et al.,
“A rational molecular design on choosing suitable spacer for better host materials in highly efficient blue and white phosphorescent organic light-emitting diodes,”
Org. Electron., 15 1368
–1377
(2014). http://dx.doi.org/10.1016/j.orgel.2014.03.028 1566-1199 Google Scholar
R. J. Holmes et al.,
“Efficient, deep-blue organic electroluminescence by guest charge trapping,”
Appl. Phys. Lett., 83 3818
(2003). http://dx.doi.org/10.1063/1.1624639 APPLAB 0003-6951 Google Scholar
X. Ren et al.,
“Ultrahigh energy gap hosts in deep blue organic electrophosphorescent devices,”
Chem. Mater., 16 4743
–4747
(2004). http://dx.doi.org/10.1021/cm049402m CMATEX 0897-4756 Google Scholar
M. H. Tsai et al.,
“Triphenylsilyl- and trityl-substituted carbazole-based host materials for blue electrophosphorescence,”
ACS Appl. Mater. Interfaces, 1 567
–574
(2009). http://dx.doi.org/10.1021/am800124q AAMICK 1944-8244 Google Scholar
T. Tsuboi et al.,
“Photoluminescence characteristics of blue phosphorescent compounds FIrpic and FIrN4 doped in mCP and SimCP,”
Opt. Mater., 31 366
–371
(2008). http://dx.doi.org/10.1016/j.optmat.2008.05.010 OMATET 0925-3467 Google Scholar
T. Tsuboi et al.,
“Spectroscopic and electrical characteristics of highly efficient tetraphenylsilane-carbazole organic compound as host material for blue organic light emitting diodes,”
Org. Electron., 10 1372
–1377
(2009). http://dx.doi.org/10.1016/j.orgel.2009.07.020 1566-1199 Google Scholar
M. H. Tsai et al.,
“Highly efficient organic blue electrophosphorescent devices based on 3,6-bis(triphenylsilyl)carbazole as the host material,”
Adv. Mater., 18 1216
–1220
(2006). http://dx.doi.org/10.1002/adma.200502283 ADVMEW 0935-9648 Google Scholar
S. H. Kim et al.,
“Deep blue phosphorescent organic light-emitting diodes using a Si based wide bandgap host and an Ir dopant with electron withdrawing substituents,”
Thin Solid Films, 517 722
–726
(2008). http://dx.doi.org/10.1016/j.tsf.2008.08.156 THSFAP 0040-6090 Google Scholar
S. O. Jeon et al.,
“Phenylcarbazole-based phosphine oxide host materials for high efficiency in deep blue phosphorescent organic light-emitting diodes,”
Adv. Funct. Mater., 19 3644
–3649
(2009). http://dx.doi.org/10.1002/adfm.200901274 AFMDC6 1616-3028 Google Scholar
H. S. Son, C. W. Seo and J. Y. Lee,
“Correlation of the substitution position of diphenylphosphine oxide on phenylcarbazole and device performances of blue phosphorescent organic light-emitting diodes,”
J. Mater. Chem., 21 5638
–5644
(2011). http://dx.doi.org/10.1039/c0jm03427d JMACEP 0959-9428 Google Scholar
S. O. Jeon et al.,
“External quantum efficiency above 20% in deep blue phosphorescent organic light-emitting diodes,”
Adv. Mater., 23 1436
–1441
(2011). http://dx.doi.org/10.1002/adma.201004372 ADVMEW 0935-9648 Google Scholar
S. H. Jeong and J. Y. Lee,
“Dibenzothiophene derivatives as host materials for high efficiency in deep blue phosphorescent organic light emitting diodes,”
J. Mater. Chem., 21 14604
–14609
(2011). http://dx.doi.org/10.1039/c1jm12421h JMACEP 0959-9428 Google Scholar
S. H. Jeong et al.,
“Comparison of bipolar hosts and mixed-hosts as host structures for deep blue phosphorescent organic light emitting diodes,”
Chem. Asia J., 6 2895
–2898
(2011). http://dx.doi.org/10.1002/asia.v6.11 CAAJBI 1861-4728 Google Scholar
J. A. Seo, M. S. Gong and J. Y. Lee,
“Thermally stable indoloacridine type host material for high efficiency blue phosphorescent organic light-emitting diodes,”
Org. Electron., 15 3773
–3779
(2014). http://dx.doi.org/10.1016/j.orgel.2014.10.020 1566-1199 Google Scholar
Y. X. Zhang et al.,
“Spiro-fused N-phenylcarbazole-based host materials for blue phosphorescent organic light-emitting diodes,”
Org. Electron., 20 112
–118
(2015). http://dx.doi.org/10.1016/j.orgel.2015.02.014 1566-1199 Google Scholar
B. Pan et al.,
“A simple carbazole-N-benzimidazole bipolar host material for highly efficient blue and single layer white phosphorescent organic light-emitting diodes,”
J. Mater. Chem. C, 2 2466
–2469
(2014). http://dx.doi.org/10.1039/c3tc32388a JMCCCX 2050-7534 Google Scholar
D. R. Lee, C. W. Lee and J. Y. Lee,
“High triplet energy host materials for blue phosphorescent organic light-emitting diodes derived from carbazole modified orthophenylene,”
J. Mater. Chem. C, 2 7256
–7263
(2014). http://dx.doi.org/10.1039/C4TC00857J JMCCCX 2050-7534 Google Scholar
C. W. Lee and J. Y. Lee,
“Structure-property relationship of pyridoindole-type host materials for high-efficiency blue phosphorescent organic light-emitting diodes,”
Chem. Mater., 26 1616
–1621
(2014). http://dx.doi.org/10.1021/cm403750p CMATEX 0897-4756 Google Scholar
M. Kim and J. Y. Lee,
“Engineering the substitution position of diphenylphosphine oxide at carbazole for thermal stability and high external quantum efficiency above 30% in blue phosphorescent organic light-emitting diodes,”
Adv. Funct. Mater., 24 4164
–4169
(2014). http://dx.doi.org/10.1002/adfm.201304072 AFMDC6 1616-3028 Google Scholar
J. J. Huang et al.,
“Novel benzimidazole derivatives as electron-transporting type host to achieve highly efficient sky-blue phosphorescent organic light emitting diode (PHOLED) device,”
Org. Lett., 16 5398
–5401
(2014). http://dx.doi.org/10.1021/ol502602t ORLEF7 1523-7060 Google Scholar
S. C. Dong et al.,
“Novel dibenzothiophene based host materials incorporating spirobifluorene for high-efficiency white phosphorescent organic light-emitting diodes,”
Org. Electron., 14 902
–908
(2013). http://dx.doi.org/10.1016/j.orgel.2013.01.012 1566-1199 Google Scholar
J. Zhuang et al.,
“Highly efficient phosphorescent organic light-emitting diodes using a homoleptic iridium(III) complex as a sky-blue dopant,”
Org. Electron., 14 2596
–2601
(2013). http://dx.doi.org/10.1016/j.orgel.2013.06.029 1566-1199 Google Scholar
S. Lee et al.,
“Deep-blue phosphorescence from perfluoro carbonyl-substituted iridium complexes,”
J. Am. Chem. Soc., 135 14321
–14328
(2013). http://dx.doi.org/10.1021/ja4065188 JACSAT 0002-7863 Google Scholar
C. Fan et al.,
“High power efficiency yellow phosphorescent OLEDs by using new iridium complexes with halogen-substituted 2-phenylbenzo[d]thiazole ligands,”
J. Phys. Chem. C, 117 19134
–19141
(2013). http://dx.doi.org/10.1021/jp406220c 1932-7447 Google Scholar
J. H. Jou et al.,
“Highly efficient yellow organic light emitting diode with a novel wet- and dry-process feasible iridium complex emitter,”
Adv. Funct. Mater., 24 555
–562
(2014). http://dx.doi.org/10.1002/adfm.201302013 AFMDC6 1616-3028 Google Scholar
S. L. Lai et al.,
“High efficiency white organic light-emitting devices incorporating yellow phosphorescent platinum(II) complex and composite blue host,”
Adv. Funct. Mater., 23 5168
–5176
(2013). http://dx.doi.org/10.1002/adfm.201300281 AFMDC6 1616-3028 Google Scholar
G. Cheng et al.,
“Structurally robust phosphorescent [Pt(O^N^C^N)] emitters for high performance organic light-emitting devices with power efficiency up to and external quantum efficiency over 20%,”
Chem. Sci., 5 4819
–4830
(2014). http://dx.doi.org/10.1039/C4SC01105H CESCAC 0009-2509 Google Scholar
H. Cao et al.,
“An orange iridium(III) complex with wide-bandwidth in electroluminescence for fabrication of high-quality white organic light-emitting diodes,”
J. Mater. Chem. C, 1 7371
–7379
(2013). http://dx.doi.org/10.1039/c3tc31365d JMCCCX 2050-7534 Google Scholar
R. Wang et al.,
“Highly efficient orange and white organic light-emitting diodes based on new orange iridium complexes,”
Adv. Mater., 23 2823
–2827
(2011). http://dx.doi.org/10.1002/adma.201100302 ADVMEW 0935-9648 Google Scholar
R. Wang et al.,
“Solution-processable iridium complexes for efficient orange-red and white organic light-emitting diodes,”
J. Mater. Chem., 22 1411
–1417
(2012). http://dx.doi.org/10.1039/C1JM13846D JMACEP 0959-9428 Google Scholar
M. Tavasli et al.,
“Colour tuning from green to red by substituent effects in phosphorescent tris-cyclometalated iridium(III) complexes of carbazole-based ligands: synthetic, photophysical, computational and high efficiency OLED studies,”
J. Mater. Chem., 22 6419
–6428
(2012). http://dx.doi.org/10.1039/c2jm15049b JMACEP 0959-9428 Google Scholar
A. Tsuboyama et al.,
“Homoleptic cyclometalated iridium complexes with highly efficient red phosphorescence and application to organic light-emitting diode,”
J. Am. Chem. Soc., 125 12971
–12979
(2003). http://dx.doi.org/10.1021/ja034732d JACSAT 0002-7863 Google Scholar
B. S. Du et al.,
“Os(II) based green to red phosphors: a great prospect for solution processed, highly efficient organic light-emitting diodes,”
Adv. Funct. Mater., 22 3491
–3499
(2012). http://dx.doi.org/10.1002/adfm.201200718 AFMDC6 1616-3028 Google Scholar
H. Fukagawa et al.,
“Highly efficient and stable red phosphorescent organic light-emitting diodes using platinum complexes,”
Adv. Mater., 24 5099
–5103
(2012). http://dx.doi.org/10.1002/adma.201202167 ADVMEW 0935-9648 Google Scholar
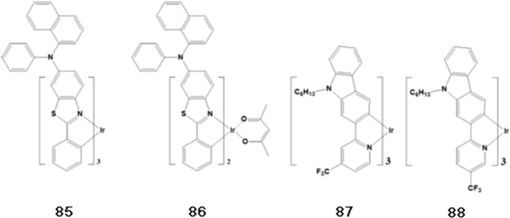
J. Moon et al.,
“ white OLEDs for solid-state lighting,”
Proc. SID, 44 842
–844
(2013). http://dx.doi.org/10.1002/j.2168-0159.2013.tb06349.x SIDPAA 0734-1768 Google Scholar
X. Xu, M. Weaver and J. Brown,
“Phosphorescent stacked OLEDs for warm white lighting applications,”
Proc. SID, 44 845
–847
(2013). http://dx.doi.org/10.1002/j.2168-0159.2013.tb06350.x SIDPAA 0734-1768 Google Scholar
H. Sakuma et al.,
“Highly efficient white OLEDs with single solution-processed emitting layer consisting of three kinds of dopants,”
Proc. SID, 44 856
–858
(2013). http://dx.doi.org/10.1002/j.2168-0159.2013.tb06353.x SIDPAA 0734-1768 Google Scholar
K. Yamae et al.,
“Highly efficient white OLEDs with over for general lighting,”
Proc. SID, 44 916
–919
(2013). http://dx.doi.org/10.1002/j.2168-0159.2013.tb06370.x SIDPAA 0734-1768 Google Scholar
M. S. Weaver et al.,
“Color tunable phosphorescent white-OLED lighting panel,”
Proc. SID, 45 672
–674
(2014). http://dx.doi.org/10.1002/j.2168-0159.2014.tb00176.x SIDPAA 0734-1768 Google Scholar
K. Hiyama et al.,
“Performance improvement of blue phosphorescent OLEDs by designing an intermolecular and interlayer combination,”
Proc. SID, 45 679
–681
(2014). http://dx.doi.org/10.1002/j.2168-0159.2014.tb00178.x SIDPAA 0734-1768 Google Scholar
J. I. Lee et al.,
“Highly efficient transparent OLEDs with an internal random nano-structured scattering layers,”
Proc. SID, 45 750
–753
(2014). http://dx.doi.org/10.1002/j.2168-0159.2014.tb00197.x SIDPAA 0734-1768 Google Scholar
T. Ishisone et al.,
“Highly efficient single-unit white OLED device with emission from both singlet and triplet excitons,”
Proc. SID, 45 762
–765
(2014). http://dx.doi.org/10.1002/j.2168-0159.2014.tb00200.x SIDPAA 0734-1768 Google Scholar
H.-K. Park,
“LG Chem develops world’s first high-efficacy OLED light panels,”
(2014) http://khnews.kheraldm.com/view.php?ud=20140911000901&md=20140914004114_BK September ). 2014). Google Scholar
|